 
So, this means that s-orbital can accommodate two electrons, p-orbital can accommodate six electrons, ten electrons in d-orbital, fourteen electrons in f-orbitals and so on. The higher the energy level, the more orbitals it has and a greater number of electrons it can accommodate. The orbitals in an atom are arranged as per the energy levels that are ordered as s, p, d and f. 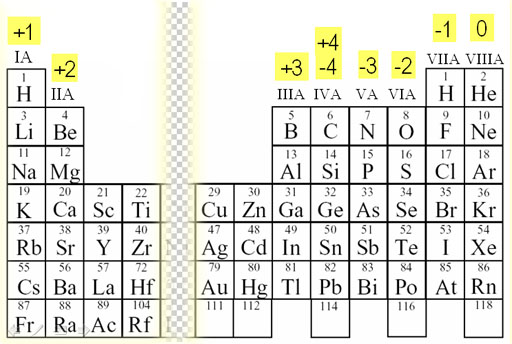
predict which element forms the most stable compounds in the +2 oxidation state. So, it is true that a magnesium ion has a net positive charge while the magnesium atom has no charge. Based on the positions of the group 14 elements C, Si, Ge, Sn, and Pb in the periodic table and the general trends outlined in this section, classify these elements as metals, semimetals, or nonmetals. Thus, whenever it loses these electrons, it becomes a positively charged ion. It has two electrons in 3s orbital and on losing these two electrons, its one shell will be removed and it will become more stable by coming close to the nucleus. We know that electronic configuration of magnesium is \. Take for example, magnesium, Mg, this has a valency of two and tends to form ions carrying two positive charges, Mg2+. When it is an atom, it has 12 protons and 12 electrons. Every atom tends to become stable by completing its octet and thus sharing electrons. The gain or loss of an electron depends on the electronic configuration of an atom. And if it gains or accepts an electron, it becomes a negatively charged ion. To use this for the -ite ions, simply subtract one oxygen but keep the charge the same. Inclusion of the word 'ate' signifies that each ends with the letters a-t-e. Number of vowels denotes negative charge quantity. When an electron is removed or lost from an atom, it becomes a positively charged ion. Number of consonants denotes number of oxygen atoms. (a) The covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. These orbits are made up of several orbitals in which electrons are placed as per their shells. Atomic radii are often measured in angstroms (Å), a non-SI unit: 1 Å 1 × 1010 m 100 pm. Atom has electrons revolving around the nucleus in their circular orbits. The term 'halogen' means 'salt-former' and compounds that contain one of the halogens are salts. They are located on group 17 of the periodic table and have a charge of -1. Because an ionic compound is not made up of single. Halogens are comprised of the five nonmetal elements Flourine, Chlorine, Bromine, Iodine, and Astatine. If an atom has an equal number of positively charged protons and negatively charged electrons, the atom is referred to as an electrically neutral atom. (Hint: Use the periodic table to predict the sign and the charge on the lithium ion.) Answer: Li2O2. An atom comprises protons, neutrons and electrons. But every atom has electrons and vacant orbitals to gain or lose electrons and become ions from atoms. The Mg ion has a charge of 2+, the sulfide a charge of 2. Every element placed in the periodic table is neutral means it has no charge when it is an atom. From the periodic table, the atomic number is the whole number that is used to put the. Hint: Magnesium belongs to group 2, alkaline earth metals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
